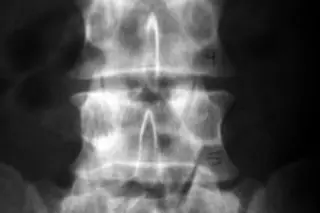
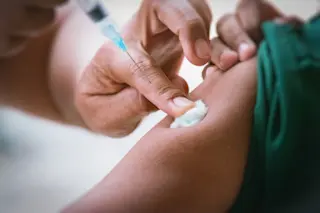
Yes, you saw a similar headline in 80beats in January of 2009, but this time we mean it. We think. Back in 2009 the FDA approved an application from Geron Corporation to begin the first human safety trials of a therapy derived from embryonic stem cells, a move that was heralded as a strong vote of confidence in this controversial but exciting area of medicine. But before the treatment of patients with spinal cord injuries could begin, the FDA reversed course and put a hold on the trial, noting that Geron had discovered cysts in some rats injected with the cells. Since then, Geron has been scrambling to prove that its treatment is safe via new animal studies, and has agreed to change some procedures to minimize the likelihood of cyst formation. Now that the FDA has signed off, Geron expects to begin the small safety trial (involving only 7 ...
FDA Green-Lights First Trials Using Embryonic Stem Cells (Again)
Geron Corporation is set to begin trials using embryonic stem cells to treat spinal cord injuries, marking a pivotal moment in medical research.
More on Discover
Stay Curious
SubscribeTo The Magazine
Save up to 40% off the cover price when you subscribe to Discover magazine.
Subscribe