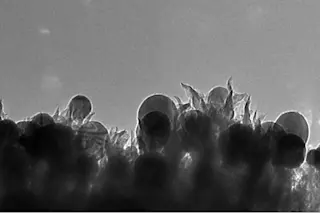
A microscopic image of the carbon nanospikes created by the researchers. (Credit: ORNL) A new chemical process turns carbon dioxide into ethanol using commonly-found catalysts and electricity. In a sense, they've figured out a way to put the genie back in the bottle. The alchemic process of converting greenhouse gases into usable energy is an appealing means of both addressing climate change and providing sustainable sources of energy. Converting carbon dioxide into energy and other useful products has been done before, but the process isn't efficient or cheap enough to implement at a large scale. Researchers from the Oak Ridge National Laboratory, however, say that they have found a chemical reaction that produces hydrocarbons using just copper as a catalyst, and they say it's quite efficient.
The key to their method is how the copper is arranged. First, the researchers create a scaffold made from carbon and nitrogen. The surface ...