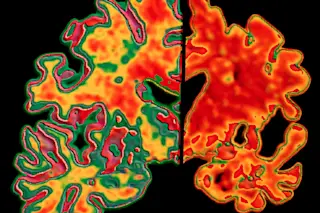
Roughly 5.7 million Americans live with Alzheimer’s, the neurodegenerative disease that slowly deteriorates memory and other cognitive abilities. For more than 25 years, many experts have pushed beta-amyloid, a protein fragment that builds up in the brain in plaques, as one of the main suspects behind the disease. Pharmaceutical companies are chasing beta-amyloid hard, racing to develop drugs to fight plaque buildup and hopefully, in turn, improve patients’ brain function. But 2018 was a mixed bag for those potential treatments.
Several companies have been developing products to inhibit BACE1, an enzyme critical to the production of beta-amyloid. But clinical trials for three of these BACE1 inhibitors ended with not-so-promising results. One was yanked for safety concerns after patients developed liver problems. Another ended early after participants had adverse effects such as trouble sleeping and suicidal thoughts, as well as no improvement in symptoms, despite lower beta-amyloid levels. The third trial ...