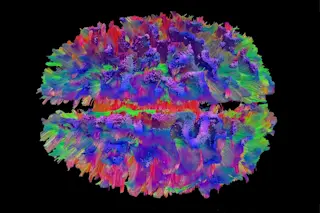
It was a frigid 17 degrees when Louis “Tom” Freund was descending a three-legged communications tower in a hayfield in Ohio. At 40 feet up, he had a splendid view of the frosted brown stalks stretching to the horizon where the cold earth met a cloudless winter sky. Tom was at the top of his game: At 42, he was running a multimillion-dollar company providing broadband Internet access to rural areas. He’d just remarried and was in superb physical shape, capable of clambering up 250-foot-high towers with 40 pounds of tools on his back, leaving colleagues half his age in his wake.
But on this day, Feb. 16, 2009, a weld snapped underfoot — something no amount of experience or physical prowess could have prevented. “I heard a loud ping, and I knew I was coming down,” he remembers.
Slicing through the icy air, he watched as the tower toppled away from him. He rode it part of the way down and at the last minute, twisted, catlike, to avoid being crushed. His aerial acrobatics saved his skull from smashing into the steel girders a moment later. The tower hit the ground, then he hit the tower, his chest smashing onto the icy metal frame. His shoulder and the right side of his head slammed into the ground. Even though a bright white haze clouded his vision, he remained conscious. “All I heard,” Tom explains slowly, “was a freight train siren going off in my head. It was deafening.”
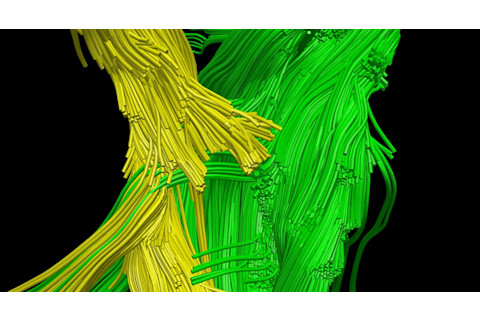
An image shows broken connections in the brain of a man injured in an ATV crash. Broken fibers (yellow) contrast with an intact area (green).Walter Schneider Laboratory
The impact shattered his pelvis and his shoulder, broke the ribs on his right side and damaged his spine and neck. Two lobes of his lungs exploded. As he lay on the frozen ground, which served as a big ice pack, a local medic called to the scene by Tom’s assistant gave him oxygen until a helicopter flew him to Allegheny General Hospital in Pittsburgh. He faded in and out of consciousness. The medics forced him to talk to his wife and kids over the radio, convinced he wouldn’t survive.
At the hospital, doctors used a CT scan to search for brain bleeds and fractures, which appear white against the fuzzy gray brain structures. Miraculously, the scan was negative. After just four days in the hospital — during which doctors successfully stopped Tom’s internal bleeding from punctured lungs and set his broken bones — he was discharged. The rest of his recovery, the doctors told him, was “an orthopedics job.” They said his brain was fine.
But Tom didn’t feel fine. He was confused, like he was in “the middle of a whirlwind,” with thoughts whizzing by that he had to grab before he could verbalize them. His sense of time was distorted. He suffered from raging headaches. His vision blurred as the day wore on, and a siren wailed in his head. He was at a constant loss for words and could not edit his thoughts, often blurting out inappropriate comments. Tom was angry, in pain, and couldn’t think clearly.

Walt Schneider (left) and neurosurgeon David Okonkwo teamed up to develop imaging that creates a wiring diagram of the brain’s neurons.Schneider: Scott Goldsmith; Okonknwo: University of Pittsburgh Medical Center
To this day, he says, his short-term memory is “nil to none,” and as we speak, he warns me that he probably won’t have any recollection of our conversation. “I don’t remember people that I’ve met a week ago,” he says.
For the first three years after the accident, Tom and his wife, Karen, searched for a plausible diagnosis. He visited a series of neurologists, each of them assuring him that he had not sustained brain damage. Next, he sought out psychiatrists and psychologists who told him he had post-traumatic stress disorder (PTSD) — extreme anxiety attacks that recur after a traumatic event. But neither he nor Karen, an emergency room trauma nurse, thought the diagnosis fit.
In the end, it wasn’t a doctor who finally identified what was wrong. An attorney who was handling Tom’s civil case suggested he had suffered a traumatic brain injury (TBI). He’d seen the same symptoms in football players he’d represented in court. It happens when the head is bumped or struck, like when a football player — or a man plummeting from 40 feet up — hits the ground. Jolts (like in a car crash) or exposure to a blast (which has affected countless soldiers who served in Iraq and Afghanistan) can also cause TBI.
These kinds of injuries can snap fragile nerves in the brain that carry signals from one part of the body to another. But there is no diagnostic technique that can visualize which nerve fibers, or neurons, are broken. When a nerve snaps, communication between different brain regions is disrupted, just like a damaged circuit in a computer. Not being able to locate the damage is an enormous setback to recovery and rehabilitation for the approximately 1.7 million people who suffer TBI each year in the U.S. That number doesn’t include the more than 300,000 soldiers with brain damage inflicted during military combat between 2000 and 2014.
Often such damage is invisible on CT scans, which use X-rays to visualize blockages, bleeds, tumors and skull fractures. MRI uses radio waves to create more detailed images, revealing bleeds, tumors and crude structural damage, but it cannot detect broken nerves. Even functional MRI (fMRI), which measures brain activity by tracking blood flow, can’t detect the loss of neurons.
Like 5.3 million Americans living with TBI-related disabilities, Tom is tormented by injuries that are invisible to doctors. Robbed of his skills, he’s been unable to work, throwing him into a downward socioeconomic spiral. Friends and family have quietly retreated, bewildered by his often inappropriate and emotional behavior, unable to recognize the man they once knew.
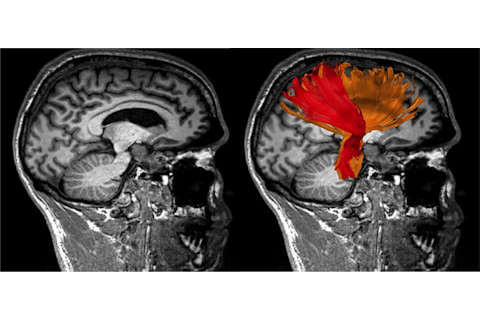
Potential injury in a patient’s brain isn’t visible with a structural MRI (left), but high-definition fiber tracking (right) reveals asymmetry in the corona radiata brain tract. The right (colored red) is smaller, consistent with the loss of motor control on the patient’s left side.Walter Schneider Laboratory
After talking with the lawyer, Karen ventually connected with a University of Pittsburgh research team working on a new brain imaging technique. In 2012, Tom set an appointment with neurosurgeon David Okonkwo, a professor of neurological surgery and the clinical director of the university’s Brain Trauma Research Center. Brain and spinal cord injuries are his specialty. Okonkwo scheduled a two-hour brain scan for Tom in a high-powered MRI machine. He then asked him to return a couple of weeks later for the results.
Neurons in High-Def konkwo knew that brain injuries were easily overlooked. Even today, when a head injury or coma patient is brought to the ER, the person gets a CT scan to determine if there is a blood clot in the brain that requires surgery. The problem, says Okonkwo, is that in nine out of 10 cases, those patients have a normal CT scan and are told they’re fine. “But in many cases, they are not normal,” he adds. “And they will be the first ones to share with you three months, six months later, the ways in which their life has changed.” He says that in most hospitals, trying to diagnose a TBI is pretty much like trying to find a bone fracture before X-ray machines were invented.
But that changed for Okonkwo in fall 2009 when Walt Schneider, a lanky, snowy-haired psychologist, visited from across campus. Schneider is fascinated by technology, and he’d come to talk about a new way to image the major tracts of the brain. Tracts are bundled cables of axons that link one region of the brain to another — like superhighways — and conduct information. An axon is the long, skinny “tail” of a nerve cell, or neuron, that transmits electrical signals from one neuron to another elsewhere in the brain. Within a specific tract, all the nerve cells begin in the same location and end in a common location. Each tract has a predominant function: The corticospinal tract controls movement; the cingulate tract, memory; and the arcuate handles language. When an axon is injured, communication between particular neurons is lost; when an entire tract is severed, two brain regions can no longer talk to each other.
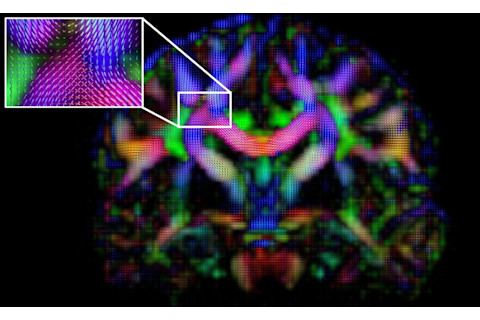
A diffusion spin diagram reveals how water molecules move in a human brain. Restrictions in the motion and speed of the molecules give researchers clues to identify larger brain tracts, or bundles of axons that link one brain region to another. The inset shows an intersection of three major tracts.Walter Schneider Laboratory
Schneider wanted a type of imaging that could produce a wiring diagram of all the neurons in the brain. But the current technology — diffusion MRI, also called diffusion tensor imaging (DTI) — didn’t allow him to see injured axons that might explain the problems of TBI patients.
Traditional DTI uses magnetic pulses to tag water molecules in the nerve cells of the brain and then records six characteristics of how these water molecules behave. The measurements are used to build images that trace the shape and direction of the axons and how the neurons in one region are connected to other brain regions. But DTI imaging had a major glitch: When axons from different tracts intersected and crossed en route to their target, the DTI software got confused and couldn’t determine each tract’s direction with complete accuracy. Schneider needed a technology that could follow these tracts from beginning to end.
With financing from the Defense Advanced Research Projects Agency, Schneider launched the 2009 Pittsburgh Brain Competition to lure the best minds to work on brain connectivity mapping. He offered $10,000 to anyone who could use data from a one-hour MRI scan to create a detailed image of the optic radiations — brain structures well defined through dissections. Teams from 168 countries signed on. As the results filtered in, Schneider’s team found that most entries were just incrementally better than standard DTI imaging. But then a Taiwanese graduate student, Fang-Cheng Yeh, sent a stunning image. His work revealed Meyer’s loop, a structure in the optic radiation, which no other team had successfully visualized. Yeh collected the reward, and Schneider enticed him to come to the U.S. to study. He worked with Schneider as part of his Ph.D. studies and is now a postdoctoral researcher at Carnegie Mellon University.
Sudhir K. Pathak, a mathematician and computational bioengineer in Schneider’s lab, analyzed Yeh’s approach. The key, Pathak found, was Yeh’s use of more and sharper observations of water molecules (257 compared with the usual six) inside the nerve cells to figure out how the axons formed neural circuits in the brain.
“MRI only sees the water,” explains Pathak. “By watching how the water moves, we can tell whether it’s moving freely in all directions or if there is something restricting movement, like a nerve cell.” Pathak improved versions of Yeh’s algorithms, producing better and higher-resolution images, and then applied the approach to the entire brain to identify and map all 40 major brain tracts. Schneider and Pathak call this new method high definition fiber tracking (HDFT). Finally, to make the wiring diagram accessible, Pathak segmented and colored major pathways involved in various neural circuits in psychedelic hues.
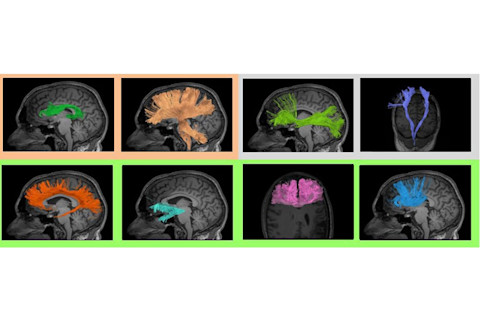
With high-definition fiber tracking, physicians can show patients the relative health of the major tracts in their brains. Colored borders are used as an aid to show those tracts with moderate injury concern (upper left), possible concern (upper right) and those within a normal range (across bottom).Walter Schneider Laboratory
Pathak spent the next year vetting the new brain images with Juan Fernandez-Miranda, a Pittsburgh neurosurgeon and neuroanatomist. He wanted to confirm that the virtual tracts he created on his computer screen matched those that the doctor saw during surgery. Fernandez-Miranda edited the images, pointing out when they were correct and when they took a wrong turn. The collaboration created a tenacious feedback cycle in which Pathak tuned the mathematics to create a tract, then Fernandez-Miranda identified what was anatomically correct. Finally, Pathak’s non-invasive virtual dissection rivaled Fernandez-Miranda’s own bloodier one.
Okonkwo immediately saw the implications and began collaborating with Schneider to test the technology in a research trial by recruiting patients with brain injuries.
Pathak and other members in Schneider’s lab then worked with Okonkwo and Fernandez-Miranda on an iPad app to create a tool that was clinically relevant and useful to neurosurgeons as they performed brain surgery or searched for damage in an injured patient. Visualizing the Damage Two weeks after the scan, Tom and Karen sat with Okonkwo in an office at the University of Pittsburgh Medical Center Presbyterian. It was September 2012, more than three years since his fall. Tom hoped to hear a conclusive diagnosis, an anatomical explanation for his troubles and the rehabilitation strategy. He had been haunted by memories of the man he was before the accident, and he longed to be himself again.
Using his iPad, Okonkwo pulled up an image of Tom’s brain. Each of the tracts was brightly colored, and looping, twisting and crisscrossing like a whorl of spaghetti.
On the screen, the left side of Tom’s brain was green and the right side, red. There tends to be a natural symmetry between the two halves of the brain, and asymmetry makes us suspicious, Okonkwo explained. Although it could be due to a natural difference between the left and right hemispheres, it might indicate an injury where circuits have been disrupted. In some regions of Tom’s brain, Okonkwo added, the circuits were asymmetrical.
He clicked on a drop-down menu and selected Tom’s Papez circuit, which is key to the control of emotions and memory. “The right side of the brain doesn’t have as much connectivity within the Papez circuit as the left side.” The right side is the one that smacked the ground.
Okonkwo explained that networked connections can be lost. If, for example, the links from the eye to the back of the brain are reduced or severed, it may diminish vision. “That concept is true for the motor system, for the sensory system, and it’s true in a slightly different way for memory, emotion, mood control,” he said. Some of Tom’s Papez circuit connections had been interrupted.
“The part of your brain responsible for encoding new memories isn’t what it once was,” said Okonkwo. He paused to let Tom digest. “And it can also be related to emotional stability and things like that.”
Karen covered her face and started to cry. For the first time, after years of doubt, anxiety and frustration, they saw the broken cables in Tom’s head. Okonkwo showed Tom another damaged brain circuit, the supplemental motor area, which is vital for integrating individual movements to make them smooth. The right side was dramatically different from the left, like someone arbitrarily hacked off huge branches of a tree. “It’s very difficult to be graceful when you have trouble with the supplemental motor area. Does that sound like you?” Tom nodded.
Okonkwo stressed that the implications of the damage were unclear; this research was in its infancy. He told them that thousands of damaged brains must be scanned before doctors understand how various injuries affect brain function.
There’s no obvious cure or therapy for Tom. But for Tom and Karen, just seeing proof that validates his symptoms felt like a step forward. “[It’s] satisfying. Sad. Scary. Heartbreaking,” said Karen. “It’s given us a confirmation that I’m not crazy,” Tom added. “For a long time, I thought I was losing my mind. Now I can finally move on.”
Seeing a detailed scan of the brain is clinically important, both in a diagnostic sense as well as a therapeutic one, says Okonkwo. “There’s actually someone who believes them.”
For the past four years, Schneider and Okonkwo have been tweaking the technology. While they are enthusiastic and hopeful about their approach, others are more cautious. Arthur Toga, director of the Laboratory of Neuro Imaging at the University of Southern California, says there are still many unknowns when it comes to the brain, questions that he and others are trying to answer as part of the national brain mapping initiative called the Human Connectome Project. Many brain circuits are not symmetrical, and simply comparing the right and left halves to detect brain damage may not be reliable. He is also concerned that telling someone the degree of damage may not be helpful. “We don’t know whether it is possible to recover those connections with the right treatments and rehabilitation strategies,” says Toga.
“Walt’s work is really promising, but it’s definitely controversial,” says Peter Bandettini, who specializes in fMRI as director of the fMRI facility at the National Institute of Mental Health. Others in the field doubt whether Schneider’s methods can truly quantify damage to specific fiber tracts, he says. For example, can his approach really determine that 78 percent of the fibers in a particular tract have been destroyed? “The jury’s still out on that.”
But Bandettini supports Schneider’s approach. “It’s important for Walt to thrash this out, push the technology and see what it can do . . . [and] he’s one of the few in brain imaging who is collaborating with the military and TBI doctors on clinical applications.”
“Actionable Intelligence” Part of that process is building the technical infrastructure that will allow Okonkwo and Schneider to better acquire MRI data, analyze and interpret it, and present brain images to clinicians and patients in a way that’s intuitive. The scan now takes 22 minutes, the analysis just four hours.
Currently the only way to get a high-definition scan of brain fibers is to participate in a research trial. That will remain true for the next three to five years until the FDA approves the technology. But already, Okonkwo and Schneider are glimpsing the fruits of their efforts: They’re helping patients understand the consequences of their brain injuries.
Treatment has been a national priority after military service in Afghanistan and Iraq resulted in a vast number of TBI and PTSD cases. Since 2007, the Department of Defense Combat Casualty Care Program has spent more than $700 million on 500-plus TBI projects, including $10 million from the U.S. Army Medical Research and Material Command for Schneider’s technology. For Col. Dallas Hack, a physician and the brain health/fitness research coordinator at Fort Detrick, Md., the advantage of Schneider’s HDFT technology is the ability to see and quantify the damaged circuits. He can use that to guide rehabilitation for the thousands of soldiers who’ve been through brain-rattling explosions.
For one 46-year-old soldier (who asked that his name not be used because of the nature of his work), participating in Schneider’s research trial has changed his life. He’s spent more than 20 years in the U.S. Army Special Forces infantry division and has served in both Afghanistan and Iraq. After surviving some 400 explosions, he had memory lapses and attention deficits that he knew compromised his ability to lead high-altitude parachute missions into enemy territory — his specialty.
An exam confirmed short-term memory loss, but the problem ran deeper. He used to be a voracious reader and was fluent in several languages. Now he could barely get through an email, written words lost their meaning, and the languages blended unintelligibly in his head.
After the scan, Okonkwo showed him the source of his problems. “My visual tracts that connect the brain to my eyes have taken a beating, which explained to me why I can’t read,” he says. “I’m not an idiot, I’m not completely broken. I just have these cables that aren’t working as well because a lot of them have been destroyed.”
Although a rehabilitation strategy wasn’t clear, in a moment of medical inspiration, one of Schneider’s team members recommended that he read to a beat — specifically using music, rhythm and doing something physical, such as tapping the words. They thought it might possibly retrain his brain to use other intact pathways.
He had nothing to lose. So he plugged in his headphones, set a beat and read his emails or had the computer read them as he looked at the words. It worked. When he’s reading, he says, “I look like I’m cutting a rap record. I’m in the studio, it’s me and Jay-Z, and we’re getting it done!” That said, it takes longer, requires technology, and it’s still not easy. “[But] I can read articles, emails, and I can read a book.” This is all possible, he says, because he had an anatomically accurate scan that revealed which brain pathways were still viable. With this new technology, the damage is now visible, and that’s “actionable intelligence,” says Schneider, who’s fond of military jargon. “In a decade, we may know how to repair the damage much more effectively.” Scans done during and after rehabilitation and the use of various medications will prove whether damaged nerves can be repaired — and might begin to reveal how that happens.

Tom Freund and his wife, Karen, listen as Pittsburgh researcher Schneider explains how high-definition fiber tracking helped identify damage to areas in Tom’s brain. That visualization was the “last piece of the puzzle,” Karen says, and helped them cope with changes in his behavior.Scott Goldsmith
Schneider’s brain scanning technology is the closest to deployment, says Hack. The advantage of this technique is the images can be processed on machines currently used at major VA, DOD and medical centers around the country. As the TBI study expands, Schneider expects to scan more than 1,000 patients in the next three years at major hospitals in the Defense Department’s Tricare health care program and at VA hospitals in Houston, San Diego, Tampa, Palo Alto, Calif., and Richmond, Va.
Schneider’s fiber tracking images are similar to those emerging from the federally funded Human Connectome Project, which is mapping neural networks with DTI and generating its own stunning collection of Technicolor maps. “[But] Walt is one step ahead of [that] project,” says Lawrence Wald, an investigator in the Massachusetts General Hospital-University of California Los Angeles consortium of the connectome project who also is collaborating with Schneider’s team. The Connectome project has focused almost entirely on imaging the brains of healthy adults to provide a “gold standard” baseline for understanding diseased or injured brains. But Schneider has developed tools specifically for TBI, says Wald, and he has assembled a multidisciplinary team of clinicians, neurosurgeons and neuroanatomists to vet the images and identify and interpret wiring differences that are clinically significant.
A New Connection Two years after seeing the damaged cable, Tom and Karen seem at ease. For Karen, a definitive diagnosis provided “the last piece of the puzzle,” she says. Understanding the reason behind Tom’s behavior has helped her adjust, as well as heal their marriage.
The diagnosis also has made an important practical impact. Tom has undergone more cognitive and behavioral tests and is still working with several doctors. Workers’ compensation is now picking up the medical bills, which had essentially wiped out his savings. Now getting the bills paid is not a constant fight. “Since the diagnosis, it seems they genuinely want to help me,” he says.
Karen still shares a special connection with Tom, but she admits that life has changed. “The Tom I know now is different. I have changed to coexist with him.” She now has a better sense of what she can expect from him, and that understanding has been freeing for both of them. “Even though it was bad news, understanding the damage to Tom’s brain was the answer to our prayers.”
[This article originally appeared in print as "Broken Cables."]