For more than 60 years, Robert Martensen’s lung cells replicated without a hitch, regulated by specialized enzymes called kinases. Much like thermostats that adjust the temperature in a room to make sure it’s not too hot or too cold, kinases make sure that the right number of new cells are created as old ones die. But sometime in his early sixties, something changed inside Martensen. One or more of the genes coding for his kinases mutated, causing his lung cells to begin replicating out of control.
At first the clusters of rogue cells were so small that Martensen had no idea they existed. Nor was anyone looking for them inside the lean, ruddy-faced physician, who exercised most days and was an energetic presence as the chief historian at the National Institutes of Health. Then came a day in February 2011 when Martensen noticed a telltale node in his neck while taking a shower. “I felt no pain,” he recalls, “but I knew what it was. I told myself in the shower that this was cancer—and that from that moment on, my life would be different.”
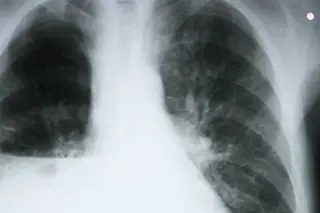
Martensen initially thought it was lymphoma, cancer of the lymph glands, which has a higher survival rate than many other cancers. But after a biopsy, he was stunned to discover he had late-stage lung cancer, a disease that kills 85 percent of patients within a year. Most survive just a few months.
Once the diagnosis was made, two questions consumed the oncologists fighting to save Martensen’s life: What was the mechanism causing the mayhem in his cells? And how could they switch that mechanism off to stop the tumor growing inside him?
Despite decades of work and billions of dollars spent to combat cancer, the odds were that no one could definitively answer either question for Robert Martensen. Every person’s cancer is a bit different genetically, making it difficult to identify its exact molecular fingerprint or to target it with drugs. That lack of precision forces physicians to give nearly all cancer patients front-line treatments, including chemotherapies, that are not only toxic but also tend to have a low rate of success. If these fail, doctors are forced into a trial-and-error approach with second- and third-line therapies, hoping to get lucky.
It seemed this would be Martensen’s fate as well. Then he heard about a new drug being tested at Massachusetts General Hospital in Boston. Developed by pharmaceutical giant Pfizer, the drug had dramatically reduced lung cancer tumors and prolonged life in the couple hundred patients who had so far used it, with few side effects. But there was a catch. The new med, called Xalkori, worked for only 3 to 5 percent of all lung cancer patients.
This poor performance is sadly typical for lung cancer treatments: Most are effective for between 3 and 15 percent of patients. The difference with Xalkori is that a test exists to identify that slim percentage of lung cancer patients (about 8,000 people in the United States and 40,000 worldwide each year) who would respond well. A regimen of Xalkori costs about $90,000 a year. But the diagnostic test, which checks to see if a patient’s tumor carries a mutation in a gene coding for anaplastic lymphoma kinase, called ALK , is just $250. And that $250 can make all the difference. Among those with a positive ALK result, 9 out of 10 saw their tumors shrink or stabilize by taking Xalkori. Most of the patients survived for at least a year; more remarkably, they were restored to such good health that they were able to return to their lives with little or no pain or side effects.
“This is what we have dreamed about: drugs that can be targeted to a patient’s specific tumor, to the mechanism that caused the cancer,” says Alice Shaw, Martensen’s oncologist and a chief investigator in the Xalkori clinical trial at Mass General.
Diagnostic tests that match up cancers with the exact drugs needed to treat them remain rare, however. “For about 80 percent of our patients with lung cancer, we don’t have tests like [the one for] ALK to tell us what treatments will work best,” Shaw says. For Martensen, the odds were slim that he had the ALK mutation that would make Xalkori effective for him. “I hoped that I might be one of the fortunate few,” he recalls. That prospect looked even more remote when Martensen’s doctors discovered that he had squamous small cell lung cancer, a less common form in which the cancer cells are scaly and thin.
Before becoming a medical historian, Robert Martensen had been an emergency room physician, a specialty whose practitioners are not known for their patience. “It was really hard to wait for the ALK test results,” he says. Yet wait he would. Shortly after Martensen’s moment in the shower, NIH surgeons removed the hard, pebble-size tumor from his neck. Pathologists embedded the tissue in paraffin to stabilize and preserve it, then cut it into sections that were layered onto slides. Finally, they stained the layers with two compounds: hematoxylin, which brings DNA into relief, and eosin, which highlights details inside cells. Those two stains enable pathologists to apply a complicated set of criteria that distinguish cancerous cells from healthy ones.
Next came the test to see if Martensen’s tumor contained the ALK mutation, which occurs when a section of DNA in the ALK gene, located on chromosome 2, breaks apart. Once the gene breaks from its natural moorings, it loses its usual ability to regulate cell growth in the lungs. With nothing to inhibit the cells from replicating, cancer is the result. No one knows what causes the gene to break apart.
To search for the mutant version of the ALK gene, technicians at NIH employed a test called fluorescence in situ hybridization, or FISH. The test uses dyes—in this case orange and green—that bind to separate halves of the ALK gene. Pathologists peer through a microscope and look for the two colors. If the two dyes are superimposed on each other, the tissue looks yellow and the gene is intact. But if the colors are separated, this indicates ALK damage. Abbott Laboratories developed the ALK -FISH test in partnership with the Pfizer team developing Xalkori. It was one of the first times that a big pharmaceutical company teamed up with a large diagnostics lab to create a cancer drug-biomarker combo.
The NIH and Mass General are at the vanguard of a major effort to identify and treat cancer based on causative gene mutations like the one found in ALK . The convergence of several factors explains the trend: cheaper genetic sequencing technologies, the discovery of new oncogenes (genes that can cause a normal cell to become cancerous), a new generation of computers and bioinformatics that can analyze vast amounts of data, and a multibillion-dollar effort by researchers inside and outside the pharma industry to develop targeted drugs and companion diagnostics for cancer.
Mass General geneticist Dora Dias-Santagata explains that she and other pathologists at the hospital’s Translational Research Laboratory examine patients’ tumors for more than 150 cancer-inducing mutations. She says they can now identify the mechanisms that cause tumor growth in half of all adenocarcinomas, a family of cancer that affects skin and other tissue, including the lungs. Adenocarcinomas account for some 40 percent of all lung cancers diagnosed. All of the known mechanisms driving adenocarcinoma in the lungs (more than eight have been discovered to date) are powered by kinase genes like ALK which, when healthy, regulate cell growth. When the genes go rogue, cells multiply out of control.
Identifying the cause of a tumor does not always mean that doctors know which drug will work best. Only a quarter of the patients who test positive for one of the oncogenes identified at Mass General can be matched to a specific treatment. “We don’t have companion diagnostics for most of these,” Dias-Santagata explains, though her lab is working hard to formulate the tests.
As the cost of DNA sequencing continues to plummet, the lab will move ever closer to a once-elusive goal: economically processing every patient’s complete genome in both tumors and healthy cells. Right now technicians target only a few crucial genes. But “probing all relevant cancer genes will mean we can find new mutations and areas of interest,” Dias-Santagata says.
A robotic device the size of a dishwasher is humming nearby as dozens of tiny tubes move tumor samples through the sequencing process. In another room of the Translational Research Lab, DNA amplifiers nicknamed John, Paul, George, and Ringo help process genes from 5,000 to 6,000 patients a year. “We’re looking for multiple mutations across tumors, mutations that turn on genes and promote cell growth,” Dias-Santagata says. “In half of patients we have found new mutations, which makes this very individualized.”
Nearby, a young technician in a white lab coat watches a computer screen for one particular patient’s signs of mutation. Anomalies appear as upward spikes on a graph line. “This is for melanoma,” the technician says. She points at an uptick on the screen. “This is a BRAF mutation [associated with melanoma] right here.”
The research behind the melanoma finding and Martensen’s ALK test began in the 1970s, when a young oncologist named Dennis Slamon became obsessed with the genetics of breast cancer. He wanted to understand why 25 percent of breast cancer patients had an identifiable, unusually fatal mutation in a gene called HER2—and to find a drug that might target this gene. His ensuing two-decade quest led to the discovery and 1998 approval of the breast cancer drug Herceptin, and to a companion diagnostic test that looks for an overproduction of her2 proteins. It was the first-ever personalized treatment for cancer.
Herceptin works by blocking the receptors for the protein produced by the cancer-causing HER2 gene for those who test positive for her2 overproduction, but it does nothing for patients who are negative. Before the advent of Herceptin, women with the mutated HER2 gene had among the worst survival rates. With Herceptin, they can now do very well. (In clinical trials, HER2-positive patients receiving Herceptin and standard combination chemotherapy had a 52 percent decrease in disease recurrence compared with patients treated with chemotherapy alone.)
Herceptin and the her2 test arrived at a heady moment for biology, when the race to sequence the human genome was close to completion and optimism ran high that more biomarker-targeted cancer drugs were close at hand. But the expected rush of personalized drugs failed to materialize because the biology driving cancer turned out to be far more complex than researchers expected.
The next round of biomarker-drug combos didn’t appear until 2004, when Genomic Health, a small start-up in Silicon Valley, launched a test called OncotypeDx. Using a panel of 21 genes, this test helped physicians target which type of cancer therapy would work best for breast cancer patients. The test identifies the 25 percent of patients who are likely to benefit from chemo and the 50 percent who should get hormonal treatments only.
At the same time, the years of research were beginning to produce a deeper understanding of the mechanisms underlying different cancers, including lung cancer. Alice Shaw recalls a signal moment in 2004—just as she was finishing her oncology fellowship at MIT—when scientists discovered that mutations in a gene for epidermal growth factor receptor (EGFR) were the culprits in about 10 to 15 percent of lung cancer patients. Perhaps more important, a diagnostic test that identified the EGFR mutation was paired with the Genentech drug Tarceva. Patients testing positive registered similar response rates to those currently taking Xalkori. “This completely transformed the field of lung cancer,” Shaw recalls.
Since 2004, a wealth of new research has produced a deluge of oncology drugs in development and human trials; more than 900 are being tested today. This is good news, although research has lagged in coming up with companion diagnostics like those for ALK or EGFR—which means that many of the new drugs are still administered in a trial-and-error fashion to determine which will work for individual patients.
In 2006 Pfizer started early human testing on one of these new, targeted drugs called crizotinib (now sold as Xalkori), concentrating on a mutation of a gene called MET, implicated in several cancers, including esophageal and stomach cancer. The link between this drug and ALK and lung cancer was not suspected until 2007, when a team in Japan published a study in Nature that made the connection. “It became clear after reading the paper that crizotinib might also work in patients with the ALK mutation in lung cancer,” Shaw says.
By December 2007 Shaw had joined the clinical team testing Xalkori at Mass General and soon enrolled her first few ALK -positive lung cancer patients. “Some had failed multiple lines of treatment and were very, very sick, with advanced-stage lung cancer,” Shaw says. The drug worked almost immediately. “One woman who had been on oxygen reported that even in the first week she could breathe better.” Physicians at Mass General and several other clinical test sites enrolled a few dozen more patients that June. They, too, did remarkably well.
“Lung cancer patients are usually treated with chemo, and they can do well, but you seldom see dramatic turnarounds,” Shaw says. “It was electrifying when we saw these reactions.”
A few days after the NIH received Martensen’s tumor, the lab issued its result. “Incredibly, it was positive,” Martensen says—he was among the fortunate few with the well-defined ALK mutation. “In my case, this was even more rare since I have a different version of lung cancer than most people who were being tested on Xalkori,” he adds. That distinction meant that Martensen still might not respond to the drug. He would soon find out as he traveled up to Boston to meet with oncologist Shaw.
For most ALK -positive patients, prospects seemed good. Xalkori had worked so well in the initial tests that Pfizer had bumped up the size of the first test population to 82 patients; two further trials followed. The drug reduced or stabilized tumors in 50 percent of the patients in one study and 61 percent in another. This was enough for the FDA to approve the drug in August of last year—after just 5 years of human testing, compared with 10 years or longer for most drugs. “We wanted to get such an effective drug out to patients as soon as possible,” reports Elizabeth Mansfield, the director of personalized medicine at the FDA.
Some of the patients who received Xalkori in those early trials in 2008 are still doing well, though over time a patient’s tumor does become resistant to the drug. As a result, Shaw’s team has begun another clinical trial with a more potent ALK inhibitor—LDK378, a drug made by Novartis—that might take over when Xalkori becomes less effective.
For Martensen, the impact of Xalkori was sudden and dramatic. “On April 12 I started taking the drug,” he recalls. “Six weeks later the node was gone. MRI scans showed no sign of disease in my brain. The tumors had been knocked down by more than 80 percent.” This transformation was accompanied by shortness of breath when Martensen exercised and a temporary loss of his sense of taste, but nothing more serious than that. His cancer remained at bay for a year before it developed a resistance to Xalkori.
Martensen recently switched to LDK378, and he feels as good as he did when he first started taking Xalkori. He goes to work each day at NIH and continues writing and lecturing about medical history. “I get tired around 3 p.m., so I work more from home. But generally I feel great,” he says. This past winter he went skiing with his four-year-old godson at Lake Tahoe and followed up with a ski trip to Switzerland in the spring. “I’ve been thrilled and grateful,” he says. “I’m having a good life right now.”
Martensen’s uplifting case remains the exception. For the almost 14 million Americans with a history of cancer, there are no miracle diagnostic-drug combos, no dramatic reduction of tumors, and no extended recovery. Forced to endure the trial-and-error regimen of drugs, these patients end up taking meds that may barely work or don’t work at all, in part because efforts to find biomarkers like ALK have not been taken as seriously (or funded as lavishly) as efforts to develop the drugs themselves.
The reasons are complicated. First is the science itself: Cancer genetics has continued to be more complex than researchers hoped. And resistance has come from the drug industry, which has strong incentives to develop products that sell to as many patients as possible. “From a business standpoint, there is not much of an incentive for drug companies to produce these diagnostics,” says Randy Scott of Genomic Health. “It reduces their market. It’s a classic business dilemma.” The regulatory environment for companion diagnostics has also confused the situation. The FDA has taken years to decide how such tests should be developed and regulated—a situation the agency began to address only last year.
Most significant is an outdated mind-set regarding how drugs are developed and used, according to Scott. “We have this whole idea of one-size-fits-all diseases and drugs, but cancer is not just one molecular event.”
“One day, I hope that doctors are like weather people,” says oncologist David Agus at the University of Southern California. “They look at the clouds and the wind speed to see what’s happening. We will look at the shape of the tumor, the structure, how it grows, and we’ll see how the tumor will respond.”
Along with Xalkori, other targeted drugs are paving the way. Last year the FDA also approved Roche’s Zelboraf, which targets a mutation implicated in melanoma with unusual dispatch. The agency interrupted a Phase III trial of 675 patients with advanced melanoma to approve the drug when it became clear it was extending survival. Zelboraf targets patients who test positive for a variant of the BRAF gene, another kinase that can go haywire and cause cells to grow out of control. Half of all patients with melanoma, which kills 9,000 people a year in the United States, carry the BRAF mutation. “We’re working to find more of these markers and targeted drugs,” Shaw says.
Martensen looks to the future as well. He knows that his cancer will eventually find its way around ldk378, as happened with Xalkori. (“If you block the mutation long enough, the tumor will likely become resistant. As with antibiotics, this is still a big problem,” Shaw says.) But he is hopeful that will not happen for at least another year or two. “In the meantime,” he says, “I got some extra time with my family and friends.”
David Ewing Duncan is author of When I’m 164: The New Science of Radical Life Extension, and What Happens If It Succeeds (2012).
Cancer Genes in the Crosshairs
Diagnostic tests and personalized therapies are now targeting several key cancer genes:
• HER2 is found in 15–20 percent of women with breast cancer and also linked to stomach, gynecological, and colorectal cancers. Drugs like trastuzumab and lapatinib block the protein produced by the gene.
• KRAS genes drive healthy cell division; mutant forms lead to leukemia and lung, colon, or pancreatic cancers. The U.S. would save an estimated $600 million every year if metastatic colorectal cancer patients were tested for it before treatment. Companion diagnostics like the ResponseDX test line can identify mutations and guide doctors’ decisions about treatments such as cetuximab and panitumumab.
• A mutant BRAF gene is either inherited, causing birth defects, or appears later in life, causing such cancers as lymphoma, colon cancer, melanoma, and lung cancer. A number of diagnostic tests, including Roche’s Cobas 4800 BRAF V600 Mutation Test, can now identify the mutation. One targeted treatment is a gene-silencing drug called vemurafenib.
• HAAH, a gene that regulates calcium, boosts tumorous cell growth if it mutates in the pancreas, breast, ovary, liver, colon, prostate, lung, brain, or bile duct. Fortunately, the mutation occurs early in the life of the cancer, so tests like a serum developed by Panacea Pharmaceuticals in Japan may help identify a target early on. Though no FDA-approved therapies exist, a few are in clinical trials.
—Jennifer Berglund