New Zealand has long been known as a place for the birds — quite literally. Before people arrived 700 years ago, the archipelago hosted an idiosyncratic ecosystem, nearly free of mammals. More than 200 bird species filled a food web all their own. Rather than cows or antelopes, there was a family of flightless birds known as moa. And in place of apex predators like tigers, New Zealand had Haast’s eagle.
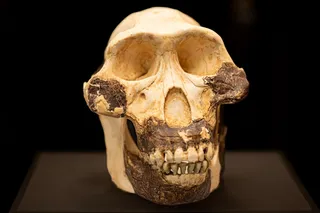
Ever since a group of farm workers drained a swamp in the late 1860s and uncovered its buried bones, this eagle has captivated researchers. Julius Haast, the explorer and geologist who published the first notes on the species, described it as “a raptorial bird of enormous dimensions.” Today, biologists estimate that the eagles weighed up to 33 pounds — roughly 50 percent more than any raptor known today. But with a wingspan of only two to three meters — ...