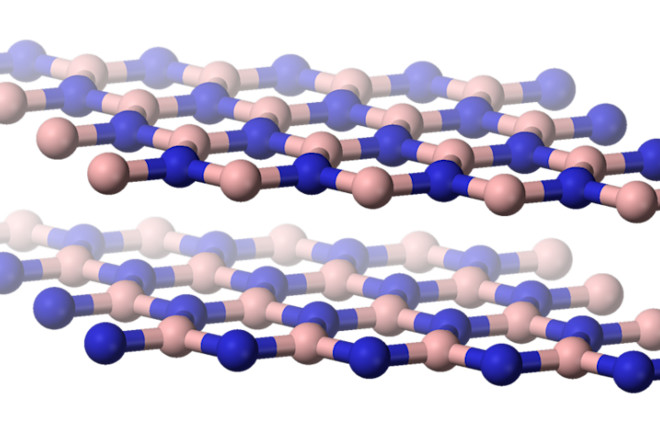
Two alternately stacked layers of hexagonal boron nitride. Nine years ago, Joshua Robinson was approached by his then-advisor with news of a discovery that would end up transforming his career, and much of materials science. “I saw this crazy talk about 2-D graphite,” he recalls his adviser saying. The adviser was referring of course to graphene, the first material to exist as truly two-dimensional: only a single atom thick. Back in 2006, the physics community was just beginning to wrap its mind around how a 2-D material could even exist. Fast forward to 2015. The realization that materials can be thinned down to the absolute limit of a single atom is spreading, both throughout the world and across the periodic table. Researchers are learning that 2-D isn’t just for the carbon atoms of graphene. Different elemental combinations can lead to fascinating new science and applications. Robinson is now associate director for Pennsylvania State University’s Center for Two-Dimensional and Layered Materials, a center with 20 faculty and over 50 students dedicated to uncovering the fundamental properties of this new zoo of 2-D materials. It is one of many such centers around the world. And as scientists continue to create new 2-D materials there’s a palpable frenzy to characterize their surprising electronic, optical, and mechanical properties. The excitement stems from the fact that materials shaved down to only a few atoms act very differently from their so-called “bulk” or 3-D version. Quantum effects begin to take hold as the electrons in the material are squeezed into that impossibly thin layer. And, being flexible, 2-D materials could bring those unique electrical properties to all sorts of new applications – from bendable touch screens to wearable sensors.
Beyond Graphene, a Zoo of New 2-D Materials Are Being Created
Newsletter
Sign up for our email newsletter for the latest science news
0 free articles left
Want More? Get unlimited access for as low as $1.99/month
Stay Curious
Sign up for our weekly newsletter and unlock one more article for free.
View our Privacy Policy
Want more?
Keep reading for as low as $1.99!
Already a subscriber?
Find my Subscription
More From Discover
Stay Curious
Subscribe
To The Magazine
Save up to 40% off the cover price when you subscribe to Discover magazine.
Copyright © 2025 LabX Media Group
