In the early 19th century, French naturalist Jean-Baptiste Lamarck put forth an evolutionary theory that the adaptations acquired by an organism during its lifetime could be handed down to its descendants. According to Lamarck, the lanky necks characteristic of modern-day giraffes, for example, supposedly emerged as a result of their ancestors' constant stretching. Though widely praised and popularized in the first half of the century, the theory was thoroughly scrutinized and debunked in the later half.
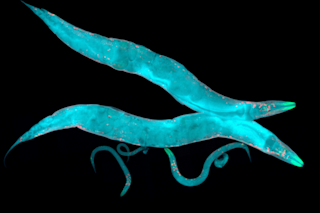
But two hundred years later, the research has come full circle. Scientists reported in Development Cell last month that parental adaptation responses to the environment can not only be handed down from generation to generation, but can even determine the course of evolution — at least in some roundworms.
Caenorhabditis elegans has become geneticists’ favorite worm in recent decades. It’s a very tiny roundworm, with a life cycle of just three days. It’s also ...