In the field of thyroid cancer, 58-year-old Kenneth Ain is a star. As director of the thyroid oncology program at the University of Kentucky at Lexington, Ain has one of the largest single-physician thyroid cancer practices in the country and more than 70 peer-reviewed publications to his name. Until recently, Ain was renowned for a highly prized repository of 18 immortal cancer cell lines, which he developed by harvesting tissue from his patients’ tumors after removal, carefully culturing them to everlasting life in vials. Laboratories around the world relied on the “Kentucky Ain Thyroid,” or KAT lines, both to gain insight into cellular changes in thyroid carcinoma and to screen drugs that might treat the disease, which strikes more than 60,000 Americans each year. In June 2007, all that changed. Ain attended the annual Endocrine Society meeting in Toronto, where Bryan Haugen, head of the endocrinology division at the University of Colorado School of Medicine, told Ain that several of his most popular cell lines were not actually thyroid cancer. One of Haugen’s researchers discovered that many thyroid cell lines their laboratory stocked and studied were either misidentified or contaminated by other cancer cells. Those included some of Ain’s. They were now hard at work unraveling the mystery.

Kenneth Ain, a thyroid cancer physician and researcher at the University of Kentucky, found that many of his cell lines were contaminated. David Stephenson
There was a disaster brewing — it just wasn’t yet official.
Ain was shocked, and justifiably so. Research based on such false cell lines would undermine the understanding of different cancers and possible treatments, and clutter the scientific literature with bogus conclusions.
“At first I thought perhaps their samples were contaminated, not mine,” Ain recalls. “So I undertook systematic thawing and genotyping of all my lines.”
He found that 17 of the 18 most frequently shared KAT lines were imposters.
It was only a matter of time before Ain figured out what went wrong. “Early in my career,” says Ain, “the head of radiation oncology at UCLA, Guy Juillard, created a number of cancer cell lines. He generously shared a few of the thyroid lines with me. And it turns out that from the beginning those original lines were likely melanoma and colon cancer.” Both grow rapidly and can easily overtake slower-growing thyroid malignancies. Human error — a researcher working on two lines at the same time, a pipette used more than once, two scientists sharing the same incubator or lab space as they worked — had likely led to the original contamination and all the lines subsequently contaminated in Ain’s and other laboratories around the country.
But rampant contamination is not the shocker in this story. Ain retired all the lines; he never sent any of them out again. He also sent letters to 69 investigators in 14 countries who had received his lines. He heard back from just two.
Across different fields of cancer research, up to a third of all cell lines have been identified as imposters. Yet this fact is widely ignored, and the lines continue to be used under their false identities. As recently as 2013, one of Ain’s contaminated lines was used in a paper on thyroid cancer published in the journal Oncogene. “There are about 10,000 citations every year on false lines — new publications that refer to or rely on papers based on imposter (human cancer) cell lines,” says geneticist Christopher Korch, former director of the University of Colorado’s DNA Sequencing Analysis & Core Facility. “It’s like a huge pyramid of toothpicks precariously and deceptively held together.” Toxicologist Thomas Hartung of the Johns Hopkins Bloomberg School of Public Health in Baltimore agrees: “Clinical drug trials in general are very well designed. They fail too often because we’re simply betting on the wrong horses in the first place. It’s a disaster.”
Like Edgar Allan Poe’s famous purloined letter, the problem of rampant laboratory contamination is out in the open for all to see, widely acknowledged by the National Institutes of Health (NIH), the National Cancer Institute, major journals and innumerable bench scientists. Yet efforts of concerned scientists have failed to stanch the tide.
“I now give regular lectures about cell line contamination,” says Ain, “and every last person in the audience is shocked and horrified. But most scientists are not willing to test and verify their lines. The NIH doesn’t require it. Very few journals require it. And I can tell you that many scientists are reluctant to disembowel their curriculum vitae, even after they find out a cell line is false. What is an ethical researcher to do?”
Too Big To Fail Cell lines are the workhorses of biology, routinely stocked and studied in every laboratory to understand cellular pathways, receptors, targets, hormones, and all aspects of normal and malignant physiology. “There is no cancer drug in current use that was not first tested in a cultured cell model,” says molecular geneticist Michael Gottesman of NIH. “Cell lines are an immortal, tissue-specific, physiologic test tube,” he says. “We need to know precisely which cell a culture represents.”

Containers of cultural HeLa cells are stored on shelves in an incubation cabinet. The cells are grown in the incubator in different concentrations in a culture medium. HeLa cells grow rapidly and have proven invaluable to all kinds of research. But their fast growth can be problematic. James King-Holmes/Science Source
When cell cultures were first crafted in 1907, they seemed to defy nature. Researchers coaxed cells to life in a broth of nutrients in hanging contraptions made of glass slides. By 1943, scientists had established the first cell line in mice.
The most studied and cherished cell line in all of biology is HeLa, cultured in 1951 from the strange, soft, purple cervical cancer of a young woman, Henrietta Lacks. The HeLa line proved extraordinarily robust. Viruses can multiply a million times in a few days in rapidly growing HeLa cells. HeLa allowed researchers to study polio, measles, papilloma virus (HPV), HIV and tuberculosis; it was used to create the first human-mouse cell hybrid, and even sent into space. It has played a role in more than 70,000 studies. HeLa is also, unfortunately, the most common cell line contaminant, responsible for more than 20 percent of contaminated cell lines. This is not news: The first widespread HeLa contamination was identified in 1967, when geneticist Stanley Gartler of the University of Washington typed 18 different human cell lines. He found that every one was actually HeLa.
The HeLa discovery was just the beginning. Work on contaminated esophageal cancer lines has led to more than 100 scientific publications, 11 patents, three NIH research grants and ongoing clinical trials involving cancer patients. The cell lines were actually lung, colon and stomach cancers. A much-studied breast cancer line turned out to be melanoma and, according to Belgian biochemist Marc Lacroix, was assumed by scientists to represent a late-stage breast cancer with an unrivaled ability to metastasize. In truth, says Lacroix, unlike the melanoma it really was, “this particular metatastic behavior is rarely seen in breast cancer progression.”
Risk of Exposure
Exposing contaminated cell lines cost Walter Nelson-Rees his career. He was an expert in culturing human and animal cells at the University of California, Berkeley, and ran a cell line bank in Oakland. From 1975 to 1981, he published a series of articles in Science outing contaminated lines and naming the laboratories where they had originated. His angry colleagues called his publications a “hit list.” In an editorial, Nature’s editor-in-chief, John Maddox, railed against “self-
Nelson-Rees’ work made it clear that HeLa contamination was far from the only problem. Eventually, the NIH terminated his contract, and he became so isolated from his peers that he left science and became an art dealer.
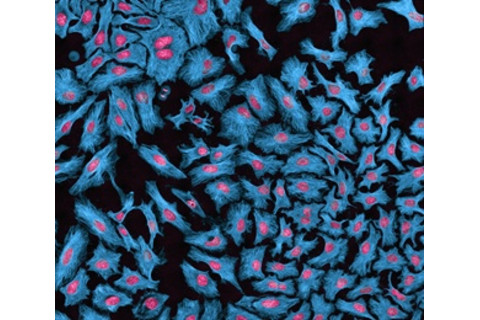
HeLa cells, with proteins labeled in blue and DNA in red, are responsible for over 20 percent of the cell line contaminations. Thomas Deerinck/Visuals Unlimited/Corbis
In 2007, a 77-year-old retired cell biologist named Roland Nardone decided to spend the final chapter of his life ending the rampant contamination. Supported by 18 other cell biology experts, he drafted a widely distributed “white paper” calling for an end to contamination. Chastened, top officials with the federal Office of Research.
Integrity sent out an email to 45,000 scientists, exhorting them to test their lines to be sure they were authentic.
By that time, the cost of validating cell lines had dramatically fallen, and the power of the testing technology had dramatically increased. A nearly foolproof form of DNA fingerprinting called short tandem repeat (STR) — sequences of DNA that are highly variable from one individual to another and therefore useful for DNA profiling — was increasingly popular. Forensic pathologists often use the technique to fingerprint DNA from blood at a crime scene. It works exactly the same way in bench science. If two cell lines share the same STR fingerprinting result, they are indeed the same. According to chemist John Butler, a former group leader with the National Institute of Standards and Technology, the chance of two cell lines coming up with the same STR fingerprinting profile is 1 in 100 million. “Scientists need to follow the fingerprinting where it leads,” says Butler. “If it’s not what you expect, it is not a problem with the fingerprinting. It’s a problem with the sample.”
But the NIH did not require fingerprinting for its grants, and so nothing changed. In 2009, immunologist Linda Miller, then executive editor at Nature, penned an unsigned editorial blasting funding agencies for allowing cell line contamination to continue. Miller suggested a global database of STR-fingerprinted cell lines. But her salvo went nowhere.
Today, cell lines known for nearly 50 years to be imposters are still in wide use under their assumed names — wrong identities regularly invoked in peer-reviewed publications. How can this be? A Sisyphean Task It’s just too damn hard.
How do you dismantle the false lines of an entire field? And just how long does it take to clean up one ordinary laboratory? Years. Ask Rebecca Schweppe, who in 2006 was recruited by endocrinologist Haugen to join the team of thyroid cancer researchers at the University of Colorado School of Medicine. Her expertise was in melanoma, which shares some features with thyroid cancer, including much-studied mutations in a gene known as BRAF.

Reprinted by permission from Macmillan Publishers Ltd John masters/Nature Reviews Molecular Cell Biology 1, 233-236 (December 2000)
Schweppe had no idea that her work was going to set off an avalanche powerful enough to reshape the field of thyroid cancer. Soon after she arrived, she ran an experiment on six different thyroid cancer lines, but her results came back impossibly clean: Three lines gave one identical result, and the other three gave another identical result. “I thought maybe I had only two thyroid lines, not six — that a number of them were misnamed and were actually redundant.”
Schweppe then turned to geneticist Korch, of the university’s DNA sequencing center. Korch was obsessed with cell line contamination. A 70-year-old Swede with silvery hair, neatly trimmed beard and gentle features, Korch has helped unmask more than 78 imposters since 2000.
Korch fingerprinted Schweppe’s lines and found that indeed there were only two. Schweppe and Haugen, worried that the contamination had occurred in their lab, asked colleagues to send them a fresh batch of the same six lines. “They turned out to be identical to our two lines,” and therefore false, says Schweppe.
But nobody yet knew what the cells actually were, since fingerprinting does not tell a scientist the origin of a cell. Without an original tissue sample to compare the cells to, the provenance of Schweppe and Haugen’s mysterious cells could remain forever uncertain. One can only pore through online databases, comparing their STR bar codes to the thousands that have already been fingerprinted, hoping to find a match.
Schweppe started searching the American Tissue Culture Collection (ATCC) database of nearly 1,200 fingerprinted human cell lines, both normal and malignant, from tissues, organs and tumors. It was as nerve-wracking as trying to find someone who had vanished into a witness protection program.
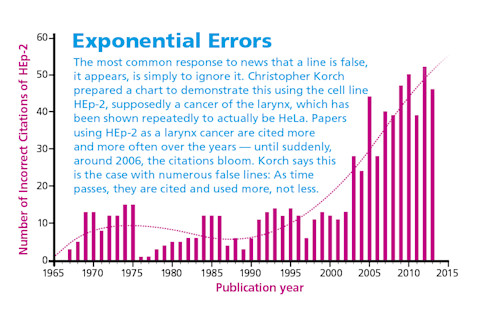
Alison Mackey/Discover after data from Christopher Korch
“One Friday at midnight,” says Schweppe, “long after my husband and kids had gone to sleep, I was at the computer once again, searching the database, and found a match for one of our lines.” The ATCC called it breast cancer, but STR fingerprinting revealed the cells were actually melanoma cells. And they were an exact match for three of Schweppe’s false thyroid cancer cell lines.
So does it matter? Yes. Those false lines were used in the early trials of two drugs that were later tested in human patients with thyroid cancer. One, called bexarotene, had no significant benefit in 17 patients after a year, according to University of Colorado School of Medicine oncologist Joshua Klopper, the study’s lead author. “I’m not sure we would have moved ahead with the trial had we found out the truth earlier,” he says.
The other drug studied, vemurafenib, also failed; thyroid cancer patients are generally resistant to the drug. “If we had still been working with melanoma cell lines, thinking they were thyroid cancer,” Schweppe says, “we wouldn’t know why thyroid cancer patients were not responding in the clinic.” Half Were False Schweppe and her colleagues fingerprinted the remaining thyroid cancer lines. In fall 2008, they reported that 17 of 40 widely used lines were imposters. During the years they were compiling their results, Schweppe would warn other scientists when she knew they were researching on false lines. But until her team’s results appeared in a peer-reviewed journal, it was difficult to get the word out. She even served as a reviewer for papers using the false lines, but couldn’t say a word. “I hated it,” she recalls.
She and the lab then won part of a $1 million grant to characterize the remaining good lines and to establish new lines from thyroid tumors. “It’s very hard to create a new line,” says Schweppe. “We worked on it for two years and produced only two new cell lines.”

Geneticist Christopher Korch, of the University of Colorado’s DNA sequencing center, has helped uncover 78 imposter cell lines since 2000. Robert Shelton
When head and neck cancer surgeon Jeffrey Myers at the University of Texas MD Anderson Cancer Center read her paper, he realized he too had published on one of the false lines. It took Myers three years to clean up his own laboratory. “I sent every cell line we had to be fingerprinted. My head technician, Mei Zhao, spent three years of her career doing nothing but unfreezing cell line stocks and sending samples of DNA for fingerprinting.” Myers tossed out 4,000 vials, and contamination forced 50 lines into retirement. “We rebuilt and fingerprinted the entire stock with more than 70 new head and neck cell lines from their original source laboratories,” says Zhao.
Even then, with a completely clean lab highly focused on meticulous technique, a new contamination struck in 2013 — something Myers identified when several different cell lines showed up with exactly the same mutation. The cause of the contamination remains a mystery. Zhao thinks the most likely possibilities are a labeling error or cross-contamination. “I was shocked to learn that contamination can even occur under close watch. Scientists need to develop standard and reliable methods to solve these issues fundamentally,” says Zhao.
Myers sees contamination as an inevitable part of science, just as errors and complications are an inevitable part of clinical medicine. “Everybody thinks, ‘Our lab is not sloppy, so nothing will happen.’ But mistakes happen in every single lab. Our field needs to establish checklists, and post them in laboratories, and make sure everybody follows them.”
Today, MD Anderson urges all its scientists to fingerprint new cell lines as soon as they arrive in the lab and before publishing any data, and to complete an annual revalidation of all lines in their laboratories. Some universities and centers have their own sequencing facilities; others send cell lines to a contractor for $30 to $80 per sample. The results come back in a few days. Sleight of Hand
In 2012, Korch was the lead author on a paper exposing 19 false endometrial and ovarian cell lines. Two of those endometrial false lines had been highly prized because they supposedly were drawn from normal tissue, which can be invaluable for studying the earliest steps to malignant transformation — potentially helping prevent cancers.
Once fingerprinting has revealed a line’s true provenance, such as HeLa, scientists sometimes claim their line does not behave or look like HeLa (or melanoma, or colon cancer, or whatever cell has invaded their line). “People treat their lines like pets,” says Gartler, who outed HeLa contamination in the ’60s. “They feel that merely by looking through a microscope they will know their cells by sight.”
But STR fingerprinting doesn’t lie. “This tool is extremely powerful and precise. It’s definitive,” he says. “If fingerprinting shows it’s HeLa, it is HeLa. Period.”
Experimental pathologist John Masters of University College London tells of a normal endothelium line that turned out to be bladder cancer, but researchers still refer to it as “endothelial-like” so they can use it in studies. (Endothelium cells line the interior of blood vessels and lymphatic vessels.) “They clearly know that these are not endothelial cells, but to get around it and not admit they are bladder cancer cells, they call them ‘endothelial-like.’ I don’t know how they reconcile the sleight of hand,” Masters says. “It is beyond my comprehension.”
Korch believes some of the waffling scientists truly are innocent: They actually don’t understand fingerprinting. They are also, he thinks, too enamored of testing cells’ behavior and function. As Korch points out, cells change appearance and function as they grow on plastic and are chemically manipulated while being studied. They also may mutate as successive generations are grown out over the years in different laboratories. Behavior is not proof of origin.
A Tarnished Reputation
In 2005, microbiologist Thomas Klonisch of the University of Manitoba created a highly prized “normal” uterine endometrial cell line, hTERT-EEC. It was a novel cell line to investigate the role of estrogen and progesterone in the endometrium, or uterine lining — including their role in endometriosis, miscarriage and cancer. “We hoped,” says Klonisch, “that researchers could use them to gain novel insights into important aspects of reproductive biology and the evolution of endometrial cancer.”

A researcher works with HeLa cells beneath a protective sterile hood, known as a laminar flow cabinet. Despite careful precautions such as these, cell contamination still regularly occurs in labs throughout the world. James King-Holmes/Science Source
Three years later, Korch’s colleague, Andy Bradford, a molecular biologist who studies gynecologic cancers, ordered three samples of the line. He sent them to Korch for fingerprinting as a matter of course. “I was very disappointed when they turned out to be breast cancer cells,” Bradford says. “Sometimes the truth sucks!” He asked Klonisch for another batch, hoping that perhaps his own lab had contaminated the first ones. But the new samples Klonisch sent were also breast cancer cells. After that, Korch and Bradford say, repeated emails to Klonisch went unanswered. During that time, research groups in at least three countries unwittingly studied and published papers on the line.
Klonisch struggles to explain why he did not immediately pay to fingerprint the line himself. One reason, he says, was the cost. At that time, the cost ranged from $67 to $475. “I offered to profile their samples as blind samples, with the identities sent to a neutral party ‘arbitrator,’ ” Korch says.
Klonisch felt he had a reasonable alternative. “We went through very extensive functional testing, and our data seemed to contradict that of Dr. Bradford. The uniqueness of our line meant that a lot was at stake. And our line did not seem to behave like breast cancer.”
How can scientists be so easily fooled by the behavior of a cell? “It’s the same concept as raising identical twins in different environments,” says Bradford. “They will look and behave differently, but their DNA remains the same.” If a breast cancer line had silently contaminated Klonisch’s cell culture early on, it would have been subject to the usual technique for immortalizing a normal cell (which involves applying enzymes, antibiotics and antibiotic-resistant genes). The only breast cancer cells that would proliferate would survive every one of those chemical manipulations. They would emerge functionally different than an untouched breast cancer cell because their environment had so radically changed.
In 2012, Korch, Bradford and colleagues published a paper detailing their false line and its breast cancer provenance. By May 2013, Klonisch did what a good scientist must: He offered corrections to the relevant journals, which have since published them. “My reputation was tarnished,” says Klonisch soberly, “and all my research in this field has been shut down. And we never intended any of this.” Winds of Change
A global correction for cell line science has begun. In 2012, Korch, Masters and 16 other scientists formed the International Cell Line Authentication Committee (ICLAC). They agreed on STR fingerprinting as the global standard for authenticating cell lines. The committee also set up a public database (found at iclac.org) of all known false lines, which numbers more than 400.
Recently, the top four cell line repositories, in America, Germany and Japan, made plans to merge their online databases of cell lines validated through STR fingerprinting, with each fingerprint converted to a searchable genetic “bar code.”
As of this writing, at least 22 journals now require cell line authentication from authors. Norbert Fusenig, the Germany-based associate editor of the International Journal of Cancer, notes that the journal has had a steady increase in submissions since 2012, when it began to require authentication. “Our impact factor, a common measure of a journal’s success, has increased,” he says.
In April 2013, Nature published more stringent requirements, in which every author had to report the source of a study’s cell lines and whether the lines had been verified recently.
Immunologist Linda Miller, whose 2009 Nature editorial called for a global database of STR-fingerprinted lines, says, “Encouraging the community to authenticate, that’s the first step, but not the final step. Ultimately, it must become a requirement. Science, and the flow of money to scientists, depends on the public trust.”
Damage Control
Scientific integrity matters more than ever, and not just for cancer research. There is a rising tide of worry over the spike in fraudulent scientific papers. • In the last 10 years, retractions of scientific papers have rocketed more than tenfold, while actual publications have increased by only 44 percent. — Nature, 2011 • The Office of Research Integrity, which pursues cases of scientific misconduct, received more than 400 allegations in 2012 — double the average from 20 years before. • In October 2013, Science correspondent John Bohannon published an article reporting a sting operation. He concocted a fraudulent scientific paper studded with anomalies and ethical approval problems, and sent it to more than 300 open-access peer-reviewed journals; more than half accepted the fake manuscript. • Of 53 papers deemed “landmark” studies over the last decade, only six held up and were reproducible. — Commentary in March 2012 in Nature by oncologist Lee Ellis • “To be successful, today’s scientists must often be self-promoting entrepreneurs whose work is driven not only by curiosity but by personal ambition, political concerns and quests for funding.” — Ferric C. Fang and Arturo Casadevall, editors-in-chief, Infection and Immunity — J.N.
[This article originally appeared in print as "Trial and Error."]