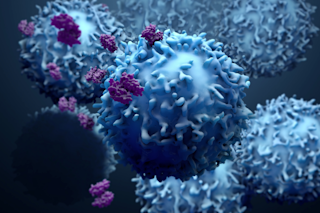
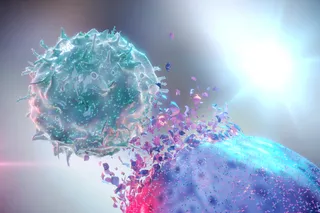
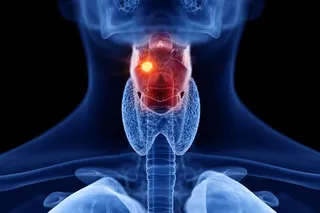
Cancer is a tricky disease to tackle. The simplicity and universality of the name belies the breadth and diversity of cancers out there. They attack every inch of the body in a myriad of ways, using a variety of tactics and DNA they can edit on the fly to evade capture and bypass our body’s defenses.
One commonality between them, though, is that they all come from within (apart from a few rare, contagious cancers like the Tasmanian devil’s facial tumor disease). They’re built from your cells. And that’s one of the greatest obstacles to treating cancers with broad-spectrum immunotherapies: It’s really difficult for your immune system to distinguish cancer cells from regular cells.
Read More: Eating Ice Cream During Chemotherapy Could Help Some Side Effects
“People have been looking for what we call cancer-specific antigens” for a very long time, says Kevan Shokat, who runs a cellular and molecular ...